Sulfide Ion Lewis Structure My XXX Hot Girl
The hydroxide ion, though, contains an octet of electrons, one more than the neutral molecule. The hydroxide ion must thus carry a single negative charge. In order to draw the Lewis structure for a given ion, we must first determine how many valence electrons are involved. Suppose the structure of H 3 O + is required. The total number of.

Lewis Dot Diagram For Sulfur Wiring Diagram
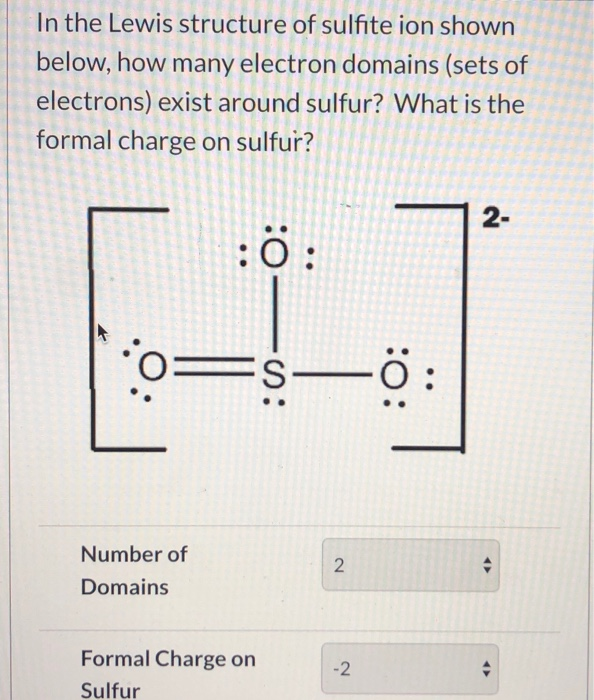
Lewis Dot of the Sulfite Ion SO 32- Back 70 More Lewis Dot Structures S does not follow the octet rule. It will hold more than 8 electrons. Sulfur having valence electrons in the 3rd energy level, will also have access to the 3d sublevel, thus allowing for more than 8 electrons.

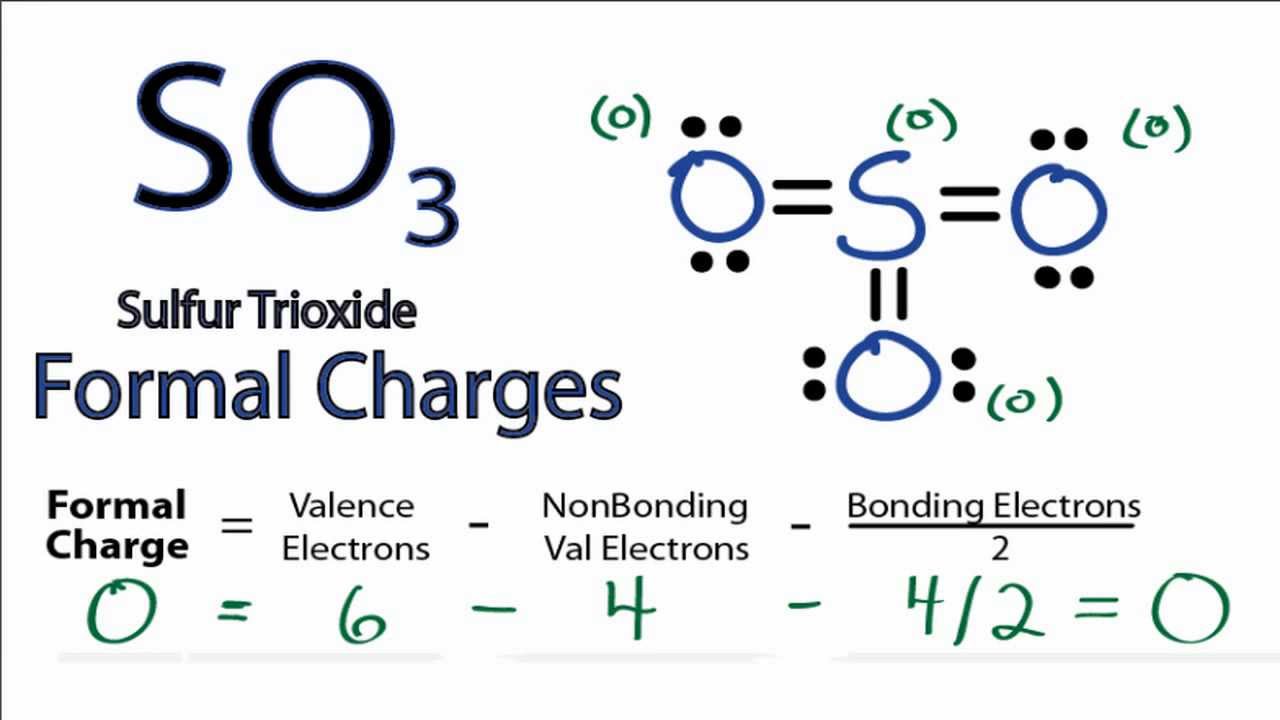
So3 Lewis Structure With Formal Charges
the greater number of valence electrons found in heteronuclear molecules. Answer. Exercise 8.9.2 8.9. 2. In molecules, as bond order increases, both bond length and bond energy increase. both bond length and bond energy decrease. bond length increases and bond energy is unchanged. bond length is unchanged and bond energy increases.
So3 схема химической связи 82 фото
Chemistry learning made easy.This tutorial will help you deal with the lewis structure and moleculargeometry of sulfite ion (SO3 2-).
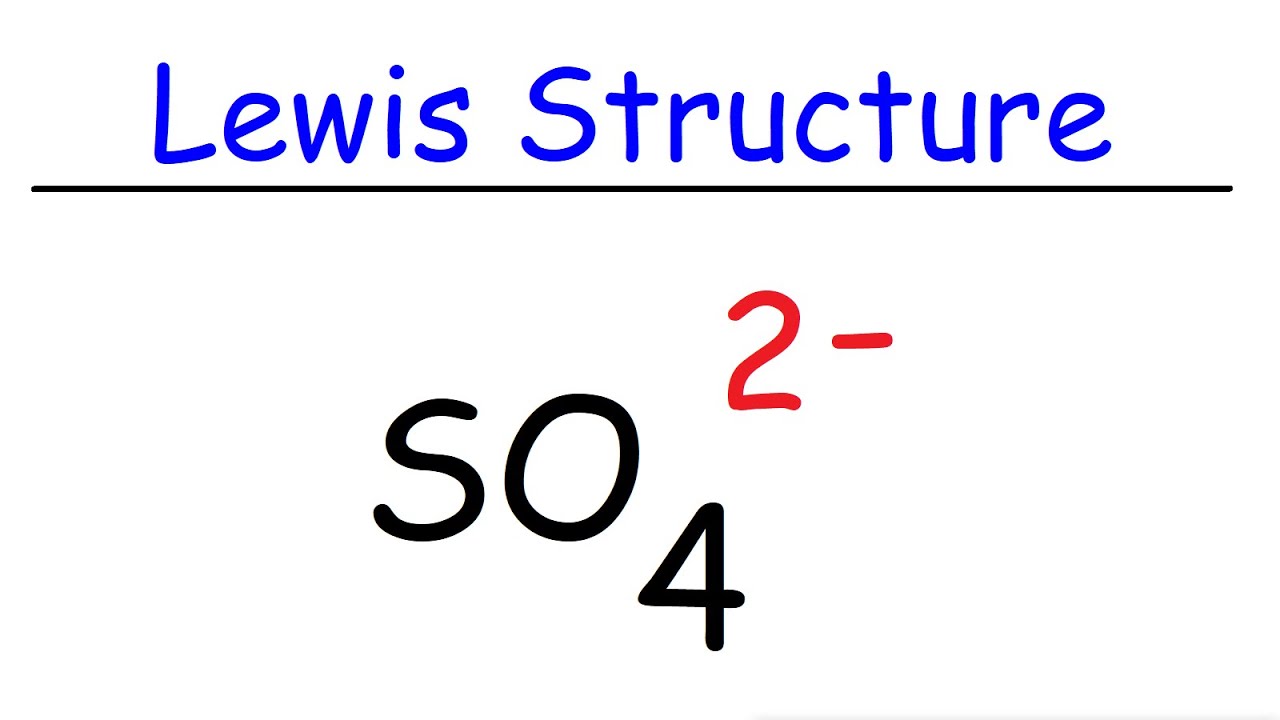
Hướng dẫn vẽ cấu trúc Lewis của so4 2 lewis structure chi tiết và dễ hiểu
Lewis structure that minimizes formal charges Lewis structure that satisfies the octet rule The sulfur-oxygen bond order is 1.33. The sulfur-oxygen bond order is 1.00. Problem 4. The Lewis structure for the ion OCN- has a single bond between oxygen and carbon and a triple bond between carbon and nitrogen.

Lewis Structure Sulfur Diagram Bohr Model Electron Hydrogen Sulfide
Sulfite ion is a weak base, but does undergo some hydrolysis to produce basic solutions. In acidic solution, the equilibria are shifted to form sulfurous acid, resulting in the evolution of SO2 gas. Sulfur dioxide is a colorless gas with a characteristic choking odor. SO2−3 (aq) +H2O(l) ↽−−⇀ HSO−3 (aq) +OH−(aq) SO 3 2 − ( aq.

FileSulfateion2Ddimensions.png Wikimedia Commons
A step-by-step explanation of how to draw the SO3 2- Lewis Structure (Sulfite Ion). For the SO3 2- Lewis structure the total number of valence electrons.

Molécula Del Anión Del Sulfito Sulfitos De Los Sulfitos Que Contienen
The sulfite ion comprises one Sulfur Atom and three Oxygen atoms. The ion has a negative.more.more Hello Guys!The sulfite ion comprises one Sulfur Atom and three Oxygen atoms. The.
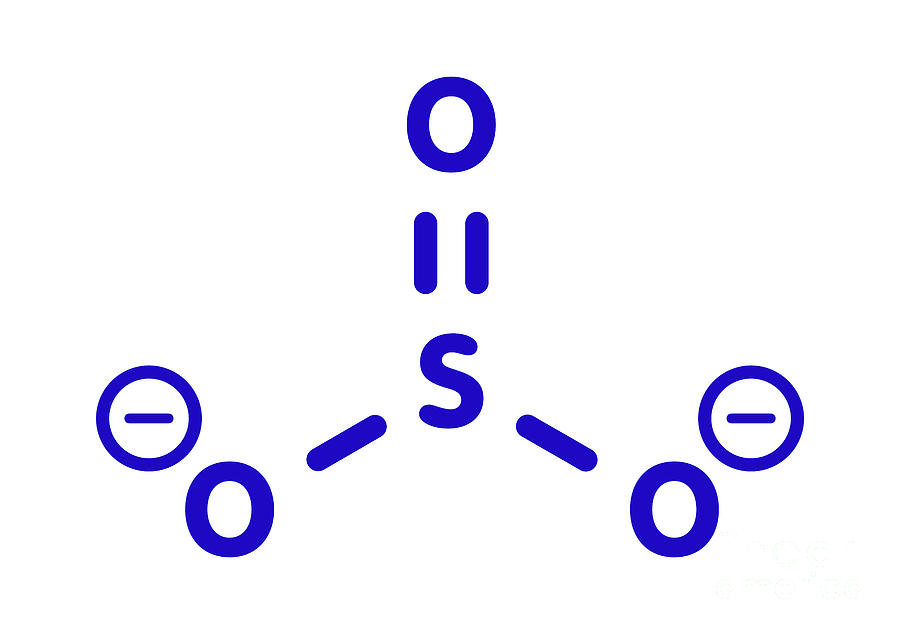
Sulfite Anion Chemical Structure Photograph by Molekuul/science Photo
The structure of the sulfite anion Sulfite is a ligand in coordination chemistry. The structure of Co ( ethylenediamine) 2 (SO 3 )N 3. [4] The structure of the sulfite anion can be described with three equivalent resonance structures.
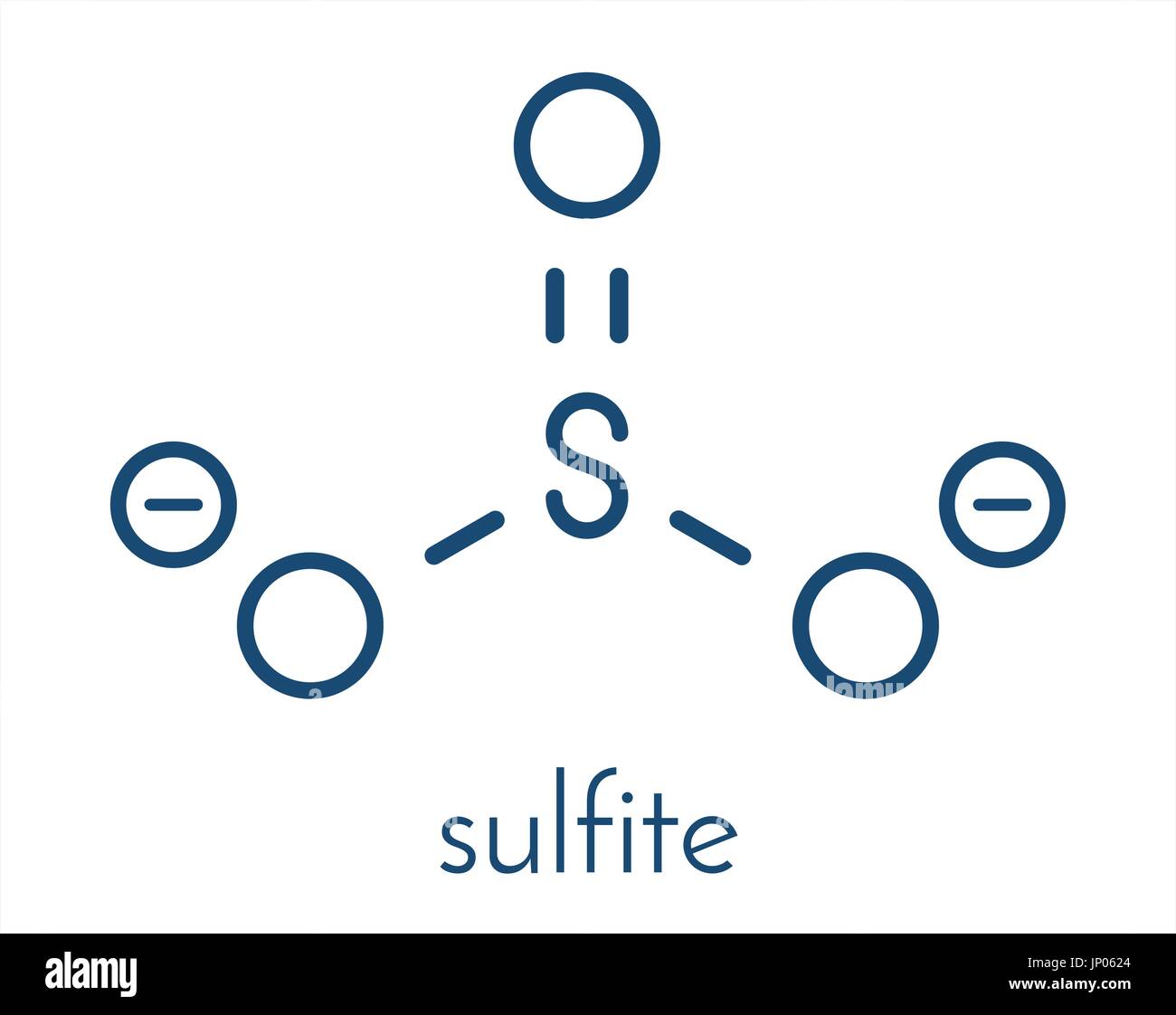
Sulfite anion, chemical structure. Sulfite salts are common food
In this video, you will learn how to draw the lewis structure for chemicals based on total valence electrons, the octet rule, duet rule, and also you will le.
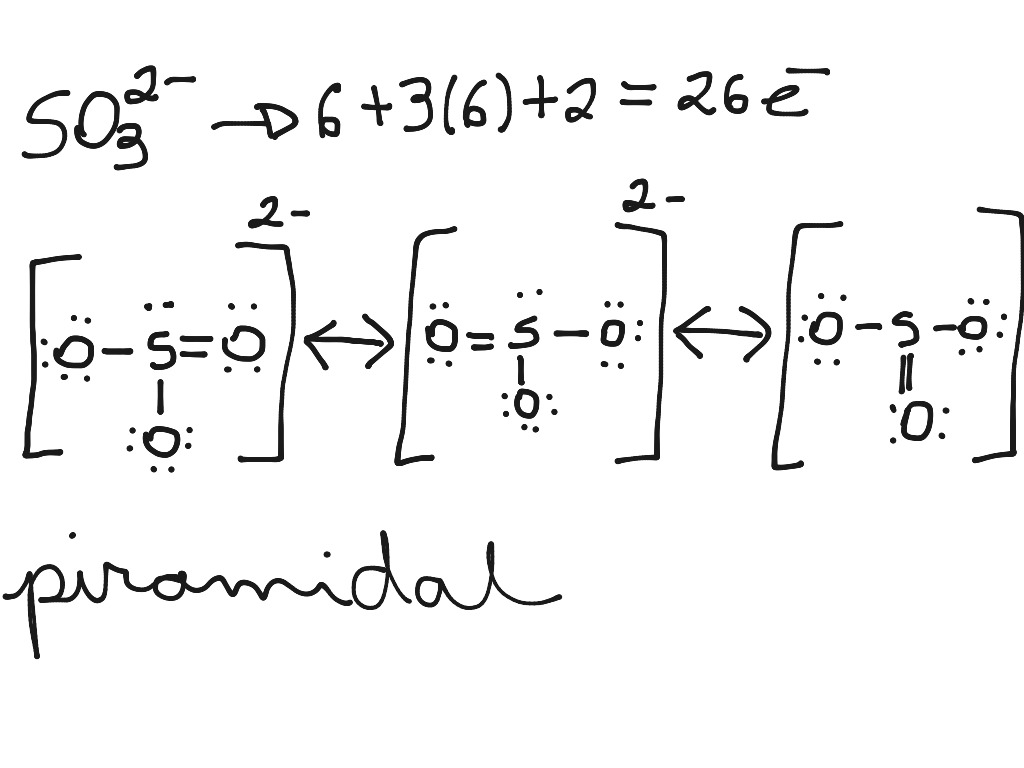
Sulfite ion Science, Chemistry, Chemical Bonds ShowMe
Lewis Dot Structure of SO3 2- (Sulfite Ion) kentchemistry.com 25.1K subscribers Subscribe Subscribed 196K views 12 years ago Every Video I quickly take you through how to draw the Lewis.

Sulfur S (Element 16) of Periodic Table Elements FlashCards
The Lewis structure of sulfite [SO3]2- ion is made up of a sulfur (S) atom and three oxygen (O) atoms. The sulfur (S) is present at the center of the molecular ion while oxygen (O) occupies the terminals, one on each side. There are a total of 4 electron density regions around the central S atom in the Lewis structure of [SO3]2-.

SO3 2 Lewis Structure (Sulfite Ion) YouTube
How to Draw the Lewis Dot Structure for S 2- (Sulfide ion) Wayne Breslyn 724K subscribers Subscribe Subscribed 461 103K views 5 years ago A step-by-step explanation of how to draw the S2-.
[Solved] Draw lewis structure for Sulfite ion SO 3 2 Course Hero
Lewis structures are preferable when adjacent formal charges are zero or of the opposite sign. When we must choose among several Lewis structures with similar distributions of formal charges, the structure with the negative formal charges on the more electronegative atoms is preferable.. In the sulfite ion,.
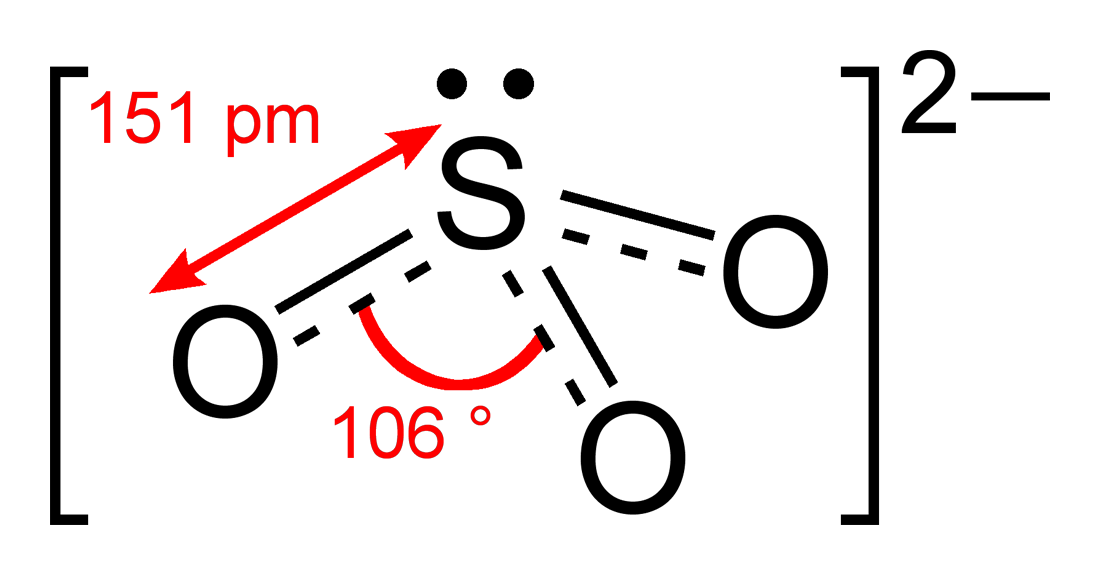
Sulfite
Video 8.5.2: Please look at Handout 1: Lewis dot structure technique while watching this video. Note steps 1 and 2 are switched but all the rest are the same. Question: What is the formal charge of each atom in the Lewis dot structure of sulfite? Answer The sulfur has a [+1] formal charge as it donates 6 electrons to the structure, but has 5.

Sodium sulfite YouTube
We are going to determine the Lewis structure for SO3 2- minus ion, also known as sulfide ion. The -2 charge you see is because it accepts two additional electrons, giving the ion a negative charge. Step-by-Step Guide to Drawing the SO3 2- Lewis Structure 1. Count Valence Electrons